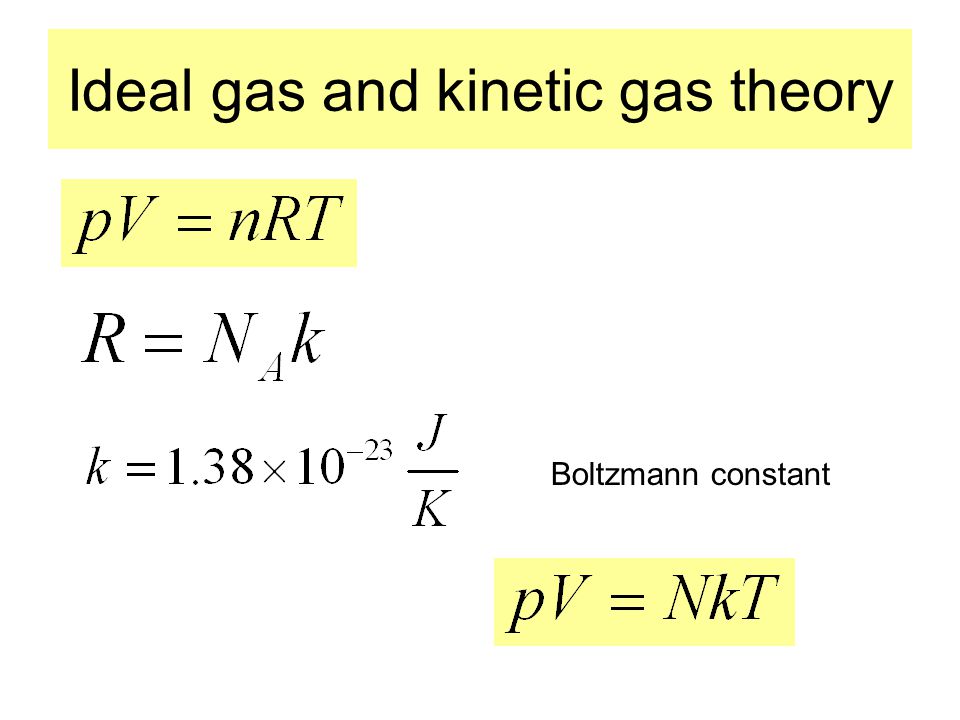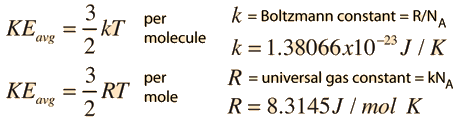The Stefan Boltzmann constant is 5.669 × 10 -14 W/mm 2 /K 4 and the... | Download Scientific Diagram

SOLVED: Boltzmann constant: kB = 1.38 x 10-28 JK Thermal energy at 300 K: kBT = 0.0259 eV Energy unit conversion: 1eV = 1.60 x 10-19 J

Twitter 上的 MathType:"The Boltzmann Constant was introduced by Max Planck in the derivation of the law of black body radiation. It is used in thermodynamics and represents the proportionality factor between the

MathType on Twitter: "The gas constant “R” is defined as the Avogadro constant “NA“ multiplied by the Boltzmann constant “k”. It is mostly known for appearing in the ideal gas law and

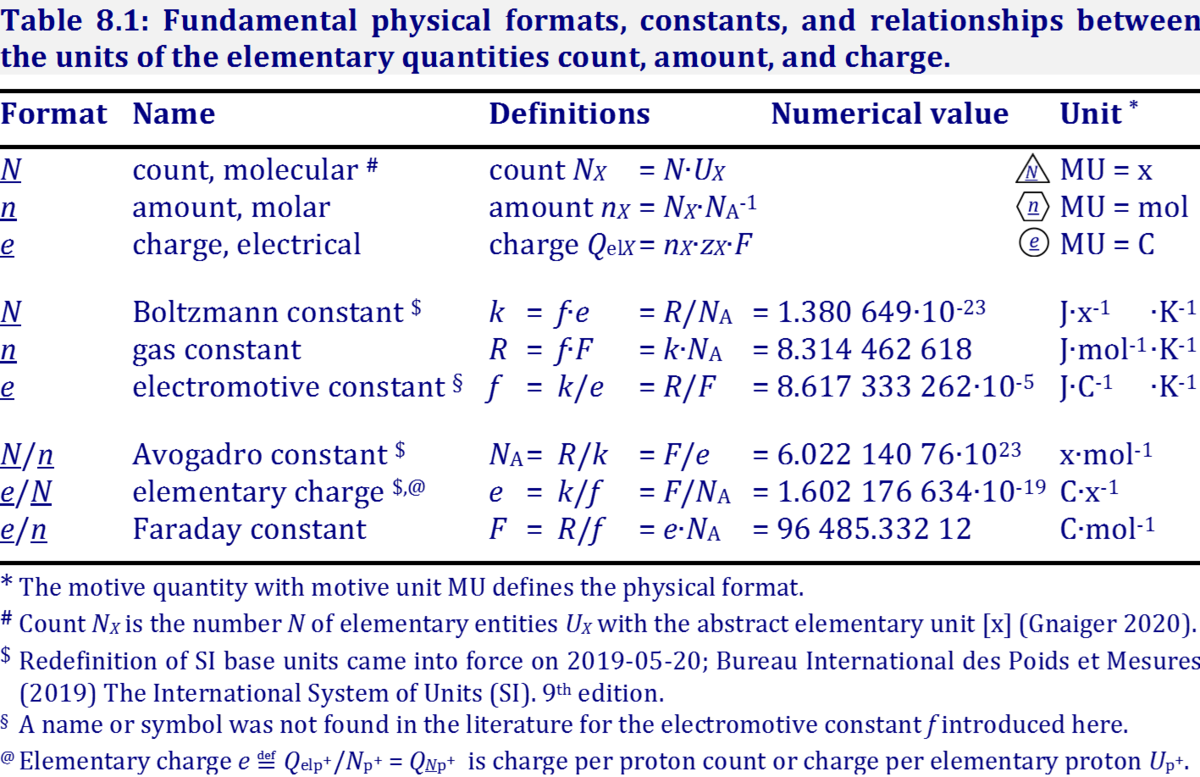
SOLVED: TABLE 13-1 Some Physical Constants and Units Used in Thermodynamics Boltzmann constant, k 1,381 X 10-2 JK Avogadro number , N 6.022 X 102: mol Faraday constant, 96,480 JWV- mol Gas