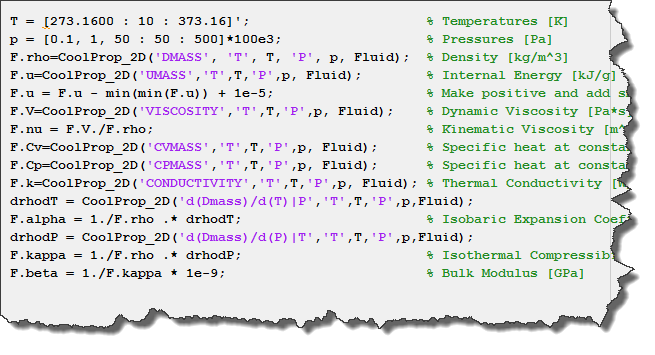
If CP and CV denote the specific heats of nitrogen per unit mass at constant pressure and constant volume respectively, then.
Ideal and Real Discharge Coefficients – Using Fundamental Equations of State in Mass-Flow Measurements with Sonic Nozzles R. S

The equation is given below, CP=CV+TV(Beta2/K), links CP and CV with Beta and K. Use this equation to evaluate CP - CV for an ideal gas. CP=CV+T(Lambda P Lambda T)V(Lambda V Lambda

The Circle of Omicron Delta Kappa, Annual Report 2022, Vol. 101, No.1 by Associate Executive Director - Issuu

![Question] Composition summation · Issue #347 · usnistgov/REFPROP-wrappers · GitHub Question] Composition summation · Issue #347 · usnistgov/REFPROP-wrappers · GitHub](https://user-images.githubusercontent.com/58614082/103503111-e8fcdf00-4e18-11eb-99f3-f320c36dd40c.png)